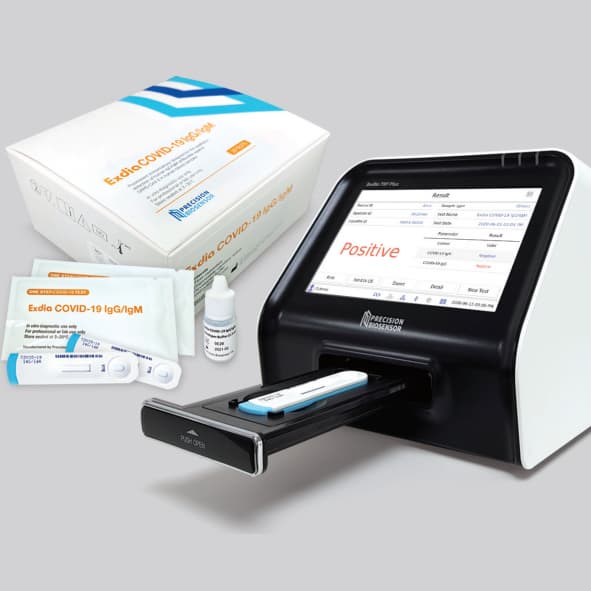
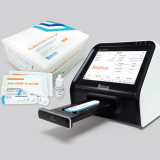
Exdia COVID-19 Antibody test & Exdia TRF analyzer
Negotiable Min Order Quantity Unit
- Required Quantity
-
- Place of Origin
- South Korea
- Payment Terms
- Negotiable
- Production method
- Negotiable
- Shipping / Lead Time
- Negotiable / Negotiable
Precision Bio Co., Ltd.
- Country / Year Established
-
 South Korea
/
2015
South Korea
/
2015
- Business type
- Manufacturer
- Verified Certificate
-
5



| Product name | Exdia COVID-19 Antibody test & Exdia TRF analyzer | Certification | CE |
|---|---|---|---|
| Category |
Examination & Testing Instrument
Monitoring & Diagnostic Equipment |
Ingredients | - |
| Keyword | antibody , rapid , covid , iggigm | Unit Size | - |
| Brand name | - | Unit Weigh | - |
| origin | South Korea | Stock | - |
| Supply type | - | HS code | - |
Product Information
ExdiaCOVID-19 IgG/IgM is a high sensitivity Time-Resolved Fluorescence immunoassay for the qualitative detection of human IgG/IgM antibodies to SARS-CoV-2 in human whole blood, plasma (Heparin/EDTA) and serum sample collected by healthcare workers at the point of care. TheExdiaCOVID-19 IgG/IgM is intended for use as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection. Test results ofExdiaCOVID-19 IgG/IgM should not be used as the sole basis to diagnosis.
Features
• High sensitivity & accuracy • Fast results in 15 minutes • For use with Exdia TRF analyzers - Objective and reliable test results - Built-in printer - Network connectivity (LIS)
Application
Serology testing for COVID-19 can be used to identify whether people were previously infected by SARS-CoV-2 even if they haven’t shown symptoms. This is important to determine, because the RT-PCR tests now being used to diagnose the coronavirus can detect the presence of the viral material during infection, but it doesn’t indicate if the person was previously infected and subsequently recovered.
• Identify asymptomatic patients • Help to verify quarantine release • Rapid screening of suspected case • Epidemiological tracing
Test procedure
1. Collect 10mL of whole blood using disposable capillary tube (provided) or 5mL of plasma or serum using liquid handling pipette (not provided). 2. Allow the disposable capillary tube or liquid handling pipette to touch lightly on the pad underneath the sample well. 3. Add two drops of developer buffer into the developer well right after injecting the specimen. 4. Read the results at 15 minutes. The test cassette should be analyzed by theExdiaanalyzer according to the instruction manual.
Clinical performance
Positive Percent Agreement (PPA) =54/57(94.74%), 95% CI: 85.63 to 98.19% Negative Percent Agreement (NPA) =145/150(96.67%), 95% CI: 92.43 to 98.57% The Overall Percent Agreement (OPA) = 199/207(96.14 %)95% Cl: 92.56-98.03%
Order information
| ||||||||||||||||||||||||||||||||||||||||||
- Verified Certificate
-

B2B Trade
| Price (FOB) | Negotiable | transportation | - |
|---|---|---|---|
| MOQ | Negotiable | Leadtime | Negotiable |
| Payment Options | Negotiable | Shipping time | Negotiable |
Precision Bio Co., Ltd.
- Country / Year Established
-
 South Korea
/
2015
South Korea
/
2015
- Business type
- Manufacturer
-
5



- President
- KIM HAN SIN
- Address
- 306 Techno 2 ro, Yuseong-gu, Daejeon, Korea
- Product Category
- Examination & Testing Instrument,Medical Test Kit,Monitoring & Diagnostic Equipment,Other Examination & Testing Instrumnet,Other Monitoring & Diagnostic Equipment
- Year Established
- 2015
- Company introduction
-
- Main Product
Related Products

Modular type ENT workstation_XU5 Visual

I-STEM (Y-STEM) PRP KIT
Covid 19 Detection
COVID 19 IgM / IgG RAPID KIT

Fetal Doppler