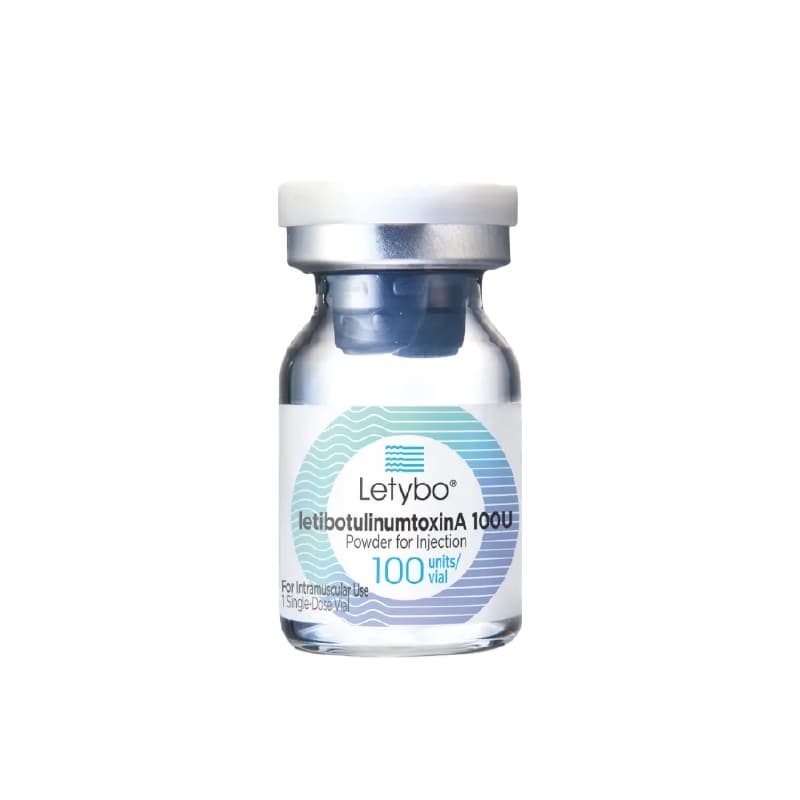
Botulinum toxin
Letybo® is a lyophilized BoNT-A product manufactured by HUGEL, a leading biopharmaceutical company in South Korea specialized in medical aesthetics.
Negotiable Min Order Quantity Unit
- Required Quantity
-
- Place of Origin
- South Korea
- Brand name
- Letybo, Botulax
- Payment Terms
- T/T
- Production method
- Available
- Shipping / Lead Time
- Negotiable / Negotiable
- Category
- Medicines

Hugel, Inc.
- Country / Year Established
-
 South Korea
/
2001
South Korea
/
2001
- Business type
- Manufacturer
Exhibition 1
Gangwon Online Beauty Show- Verified Certificate
-
3



| Product name | Botulinum toxin | Certification | - |
|---|---|---|---|
| Category | Medicines | Ingredients | LETIBOTULINUMTOXINA |
| Keyword | toxin , toxins | Unit Size | 44.0 * 36.0 * 66.0 mm |
| Brand name | Letybo, Botulax | Unit Weigh | 30 mg |
| origin | South Korea | Stock | 10000 |
| Supply type | Available | HS code | 3002 |
Product Information
The FDA approved Letybo®(Botulax®) is a lyophilized BoNT-A product manufactured by HUGEL,
a leading biopharmaceutical company in South Korea specialized in medical aesthetics.
Letybo® contains the active ingredient, letibotulinumtoxinA (~900 kDa)
- #1 botulinum toxin player in South Korea, a competitive aesthetics hub, for 7 consecutive years1
- Approved in over 60 countries, including the USA, EU, Canada, and Australia, China, Taiwan...
- Accumulated sales of over 26 million vials from 2010 to 2022
INDICATION
Temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/or
procerus muscle activity in adults.
* Details of market authorization may differ by product and by country.
LETYBO SPECIFICATION
- Active Ingredient Clostridium botulinum toxin type A
- Packing unit (1vial) 50 unit / 100unit / 200 unit
- Appearence A white injectable dry powder in a transparent glass vial
which should become a colorless, transparent liquid when reconstituted
with a solvent (normal saline solution)
- Efficacy Effect
1. The Treatment of Positive Essential Eyelid Convulsion in Adults aged 18 +
2. Temporary Improvement of Severe Las so wrinkles in Adults aged 18 to 65 years of age,
associated with Corrugator Muscle and/or Procerus Muscle activities
3. Muscle Stroke : Treatment of Upper Limb Stroke in Adults aged 20 or older
4. Treatment of Dynamic Equinus Foot Deformity in children with cerebral palsy over 2 years old.
Temporary Improvement of External Angles (Eye Wrinkles) of Secondary Ideals related to Orbicularis Oculi Muscle Activity in Adults aged 19 to 65
- Product Info Attached File
B2B Trade
| Price (FOB) | Negotiable | transportation | Negotiation Other |
|---|---|---|---|
| MOQ | Negotiable | Leadtime | Negotiable |
| Payment Options | T/T | Shipping time | Negotiable |

Hugel, Inc.
- Country / Year Established
-
 South Korea
/
2001
South Korea
/
2001
- Business type
- Manufacturer
Exhibition 1
Gangwon Online Beauty Show-
3



- President
- Hyong Jin Moon
- Address
- 17 samseongro 133 gil, Gangnam-gu, Seoul, Korea
- Product Category
- Face Cream & Lotion,Medicines,Other Pharmaceuticals,Skin Care Serum,Skin Care Set
- Year Established
- 2001
- No. of Total Employees
- 501-1000
- Company introduction
-
Hugel will take a bold leap forward to become a global top-tier company that provides health and beauty to humanity and create value to realize a happy life.
Hugel is a global leader in the aesthetics and therapeutics market dedicated to providing proven, high quality products such as botulinum toxin, HA filler, PDO thread and derma cosmetics.
Hugel drive to enter the US, Europe, and China for the first time in Korea
and recognized in more than 70 countries around the world.
- Main Product
Related Products
NOVA SlimBS
_2.jpg)
VINIX ORAL DISSOLVING FILM 50mg,100mg(SILDENAFIL 50mg, 100mg)
NOVA PMC
Medicine for dentin hypersensitivity
Novel small molecules for treating NASH